Overview
Once overlooked as a mere evolutionary vestige, the primary cilium is now recognized as the antenna that senses and conveys signals into the cell from the extracellular milieu. This microtubule-based, membrane-ensheathed organelle protrudes from the surface of most cells in our body. Its miniature volume dramatically concentrates signaling molecules for efficient and precise transmission of their messages into the cell.
Ciliary dysfunction is associated with a plethora of human diseases, ranging from kidney malfunction to cognition impairment. These diseases, collectively known as ciliopathy, implicate mutations in hundreds of genes. Remarkably, many ciliopathy patients exhibit various deficits in brain structure, suggesting that primary cilia play fundamental roles in brain formation.
We aim to elucidate the mechanistic roles of cell signaling in the primary cilium that govern neurodevelopment, and to decipher how signal transduction errors lead to birth defects and developmental disorders.
![]()
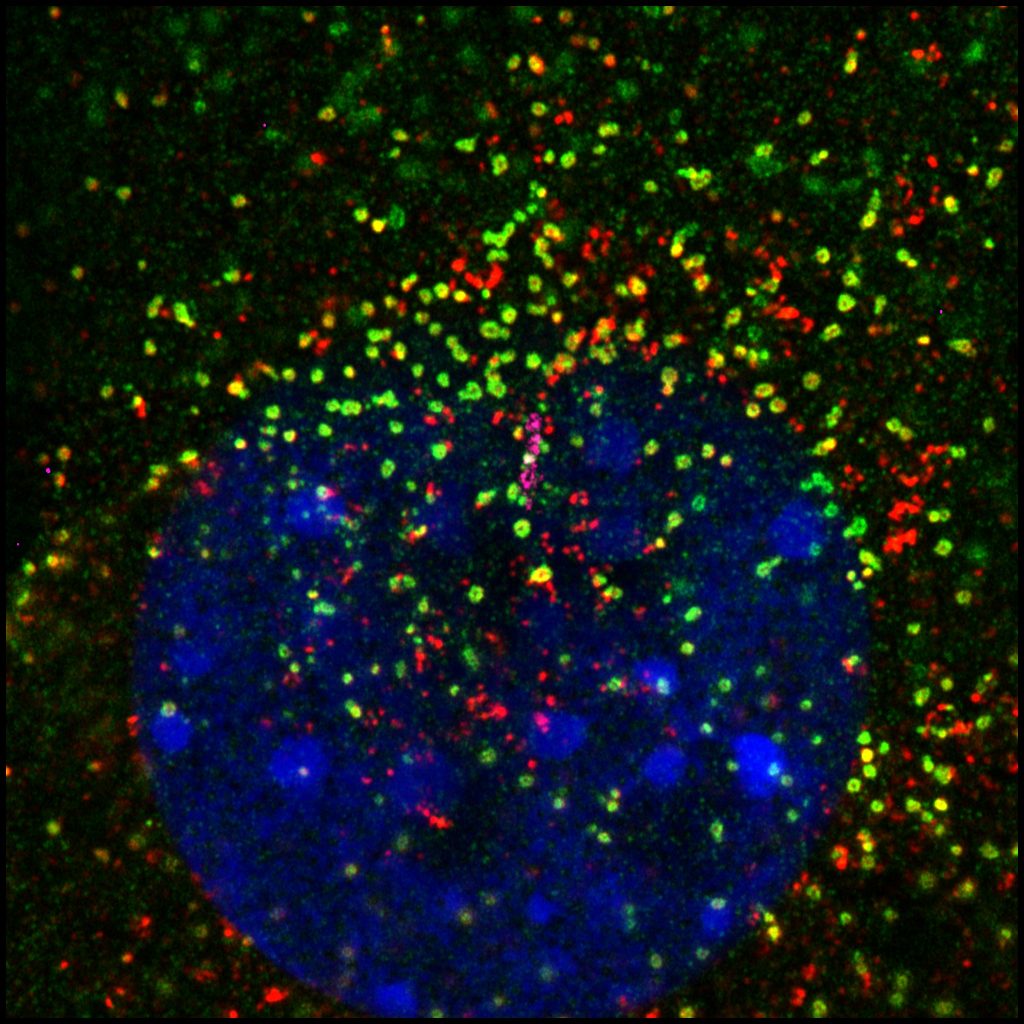 Elucidate transduction mechanisms of Hedgehog signaling in the cilium
Elucidate transduction mechanisms of Hedgehog signaling in the cilium
The Hedgehog (Hh) pathway is the first cell signaling discovered to rely on the primary cilium. Hh signaling plays fundamental roles during embryonic development and in the maintenance of adult stem cells; dysregulated Hh signaling leads to various birth defects and tumorigenesis. Yet the transduction mechanisms of many steps of Hh signaling remain mysterious to us. We aim to elucidate this process by characterizing dynamic ciliary protein transport in response to the Hh agonist. We have leveraged the proximity-labeling based enzymes and quantitative proteomics to generate the atlas of ciliary proteins before and after Hh activation. By targeting the enzyme to different microdomains of the cilium, we discovered distinct cohorts of ciliary proteins. These results opened new doors for the understanding of Hh signal transduction mechanisms.
![]()
Define roles of cell signaling in the primary cilium of the developing brain
The primary cilia of neural progenitors in the developing brain have long been viewed as the signaling hub that senses cue molecules from the cerebrospinal fluid (CSF) to govern brain formation. Yet strikingly little is known on what cell signals are sensed and conveyed from CSF to neural progenitors. This leaves a massive void in our understanding of embryonic brain development and obscures the pathological elucidation of neurodevelopmental disorders. To gain insights into this long-time enigma, we aim to systematically plot signaling proteins in the cilia of neural progenitors over space and time. We will harness the proximity labeling method established in our lab to catalog bona fide cilium proteins operating in discrete brain regions and across different developmental stages. Results from this project will chart new directions for future studies of embryonic brain development.
![]()
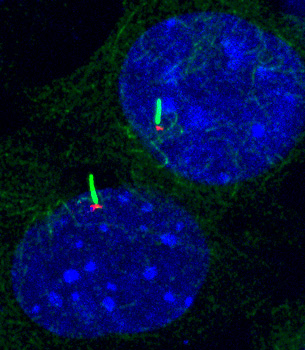 Determine regulation of Hh signaling by compartmentalized PKA in the cell
Determine regulation of Hh signaling by compartmentalized PKA in the cell
Protein kinase A (PKA) is a well-known suppressor of Hh signaling. Genetic ablation of PKA leads to maximal activation of Hh signaling in mouse embryos. How could such a housekeeping kinase selectively regulate Hh signaling? Studies in adrenoreceptors suggest that PKA activity in the cell is compartmentalized: receptors and phosphodiesterase (PDEs) within specific subcellular microdomains precisely control local PKA activity, which then accurately convey the signaling to downstream effector proteins. Our results and others show that PKA at the centrosome and in the cilium selectively governs Hh signaling. We have shown that selectively elevating PKA activity at the centrosome, via dispersing centrosomal PDE4D, suppresses Hh signaling and Hh-related tumorigenesis (Ge et al, eLife 2015; Peng et al, Mol Biol Cell, 2021). We have also found that PKA catalytic subunit binds to Smo and is retained in the primary cilium by active Smo, allowing the subsequent activation of Hh signaling (Happ et al. Nat Struct Mol Biol, 2022).
How does centrosomal and ciliary PKA coordinate to regulate Hh signaling? How does PKA command the activation of Gli transcription factors during Hh signal transduction? What is the significance of compartmentalized PKA in Hh-related tumorigenesis? We will combine advanced techniques in molecular and cellular biology, quantitative proteomics, optical imaging, and mouse genetics to shed light on these questions.
![]()
We are thankful for the following funding support:
